|











|
Rosellinia corticium (Schw.: Fr.) Sacc.
Stromata uniperitheciate, usually densely gregarious, rarely fused
into 2-3- peritheciate stroma, subglobose with a flattened to somewhat cupulate
apex, often constricted at the base, brown to blackish, 0.8-1.3 mm
high x 0.9-1.4 mm diam; ectostroma hard, carbonaceous; subiculum brown to purplish brown, abundant,
woolly, persistent at least at the base of mature stromata, long adhering to
side walls, usually conspicuous
between the stromata.
Ostioles papillate, conical.
Asci cylindrical, with apical apparatus cylindrical to more or less urn-shaped, amyloid,
7-11 µm high x 4-6.5 µm broad.
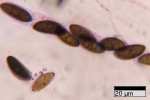
Ascospores 19-27 x 7.5-10 (-12) µm, ellipsoid-inequilateral with broadly
to narrowly rounded ends, dark brown, with a frequently sigmoid or oblique germ slit nearly
spore-length on the
more convex side; a cellular appendage usually present at one end, at times
at both ends, with a slimy sheath around the whole spore visible on fresh and
young material.
Anamorph in nature: Geniculosporium-like, grey to light brown,
present on subiculum at young stages.
Specimens examined.
FRANCE: Aričge (09): Lapenne, Parc des Bambous,
banks of Hers river, 06 Apr. 2002, JF-02066, on rotten culms of Arundo donax;
Rimont, Las Muros, 24 Sept. 2002, JF-02179, on Laurus
nobilis; Rimont, Las Muros, 19 Dec. 2002, JF-02248, on Quercus robur;
Rimont, Las Muros, ruisseau de Peyrau, 19 Dec. 2002, JF-02249, on Acer campestre,
Rimont, Saurine, 12 Feb. 2002, JF-02030, on Salix caprea.
Gironde (33),
Mongauzy, 8 Feb. 1992, JFM-9201 on Quercus or Carpinus.
Haute Garonne (31):
Fourquevaux, 26 Mar. 1995, JFM-9527, on broad leaved tree; Saleich, Artihagučre,
10 Feb. 2000, JF-00010, on Populus tremula; Toulouse, CRS park, 11 Dec. 1994, JFM-94162, on broad leaved tree.
Pyrénées Atlantiques (64):
La Bastide-Villefranche, Lac de la Pounte, 06 Nov. 2003, JF-03233, on Platanus
sp.;
Sauveterre de Béarn, 22 Feb. 1992, JFM-9210, on broad leaved tree. Sarthe
(72): La Ferté Bernard, 08 Aug. 1986, PL-860808-01, leg. Paul Leroy, on Quercus.
Vendée(85): Avrillé, Bois de la Garde, 05 Jun. 2003, JF-03090,
on Quercus.
Notes: Rosellinia corticium is macroscopically identical with
R. aquila and both species are easily confused in the field. As stated
by Petrini (1992), R. corticium is readily distinguished from R. aquila
by larger ascospores, surrounded by a slimy sheath, and asci with a larger apical
apparatus.
In fact the slimy sheath is often inconspicuous, except around immature ascospores,
and a careful exmination of fresh material shows that a second cellular appendage
is often present, although very inconspicuous.
Ju & Rogers (1999) also report variations in the presence and number of
cellular appendages in both R. aquila and R. corticium in north
American material, and consider the latter a large-spored species within the
R. aquila complex. In our experience, in large-spored specimens we name
R. corticium, the larger size of ascospores is correlated with larger
apical apparati and germ slits frequently oblique to slightly sigmoid.
The collection JF-02248 is distinctive in that most stromata exhibit a pinkish
grey ring around the flattened ostiolar area, a feature not previously reported.
Other macrocospical and microscopical features are otherwise in full agreement
with those of typical R. corticium.
Another collection (JF-02066) on Arundo donax deviates from typical
features of R. corticium in having shorter ascospores 17-20
x 6.5-8 µm and smaller apical apparati 4.8-6 µm high x 3.4-4 µm broad. Although
these characters rather recall R. aquila, this collection is tentatively
kept in R. corticium owing
to the complete slimy sheath surrounding its ascospores and presence of two
cellular appendages. In this collection, ascospores exhibit a wide range of
morphological variations, often beaked or misshapen, therefore their dimensions
may be misleading. Additional collections are needed to clear up its connections
with R. aquila or R. corticium.
Rosellinia corticium is not uncommon in our region, frequently colonizing
branches of recntly fallen trees. It is plurivorous on deciduous trees wood,
with an apparent host preference for Quercus. It has a worlwide distribution,
known from Europe, North and South America (Brazil, Mexico) and Asia (India,
Taiwan) (Petrini, 1992; San Martin Gonzalez & Rogers, 1995; Ju & Rogers,
1999). The absence of records of R. corticium from British
Isles (Petrini, 1992; Rogers & Ju, 1998) where it seems "replaced"
by R. aquila, is noteworthy but needs further
confirmation.
|